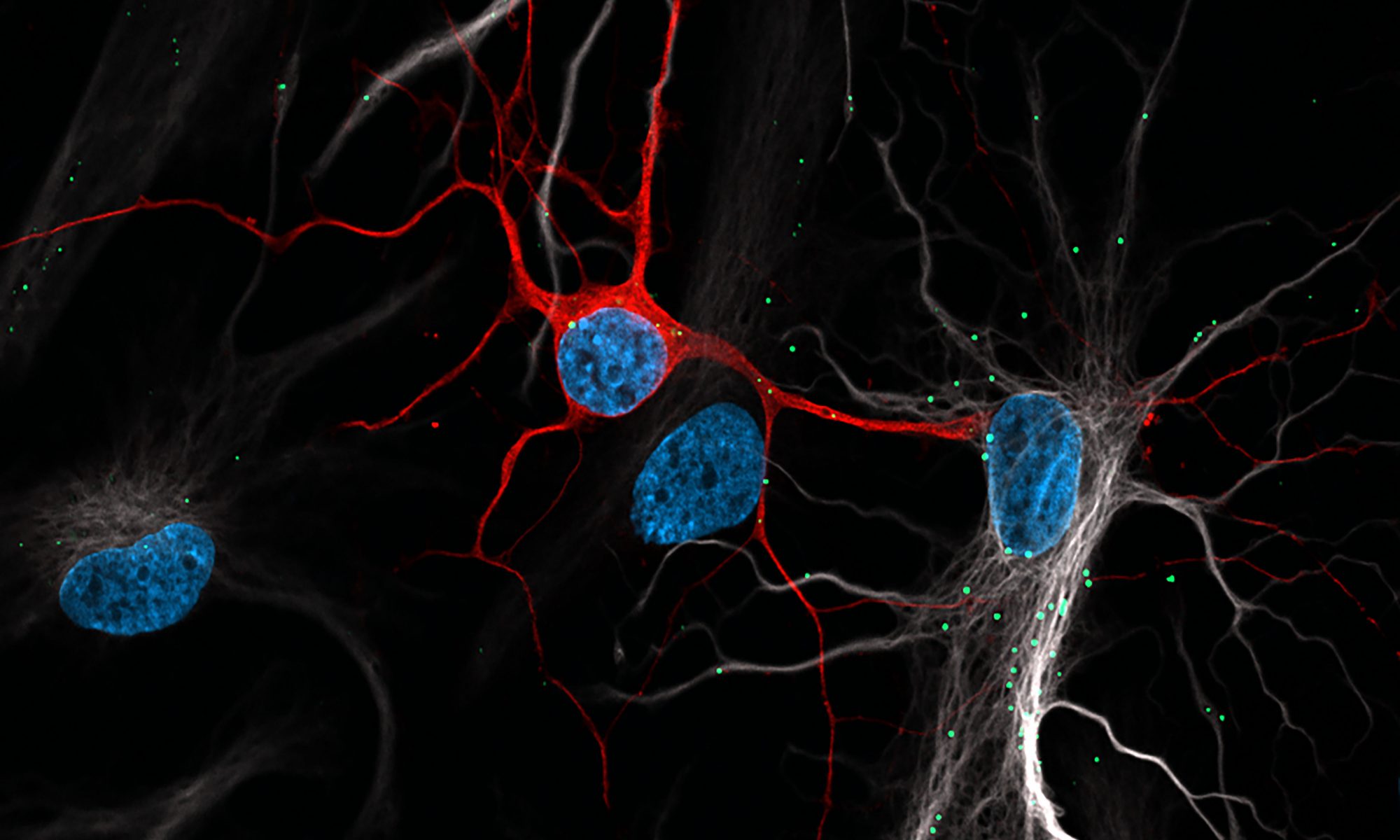
Seeing is believing. Our lab uses a combination of advanced quantitative microscopy techniques to study lipid trafficking in the brain. We use live cell imaging to understand the dynamic nature of our trafficking pathways, super-resolution microscopy to resolve sub-diffraction structures and high-sensitivity confocal microscopy to study organelles in tissue. As model systems we use primary neuron and glial cultures, biochemical assays and in vivo mouse models of stroke.
1. Lipid trafficking in the brain
While glia have long been known to supply neurons with lipids and cholesterol via lipoprotein particles, new studies show that neurons transport excess lipids back to glia in an attempt to avoid lipotoxicity following oxidative stress. Our lab seeks to uncover the unique molecular machinery used in this process.



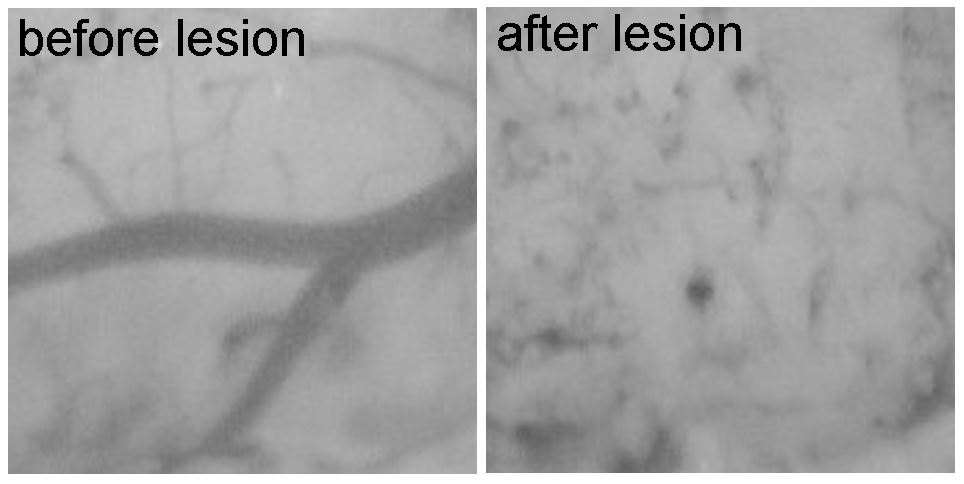
2. Lipid droplet formation in neurodegenerative disease and stroke
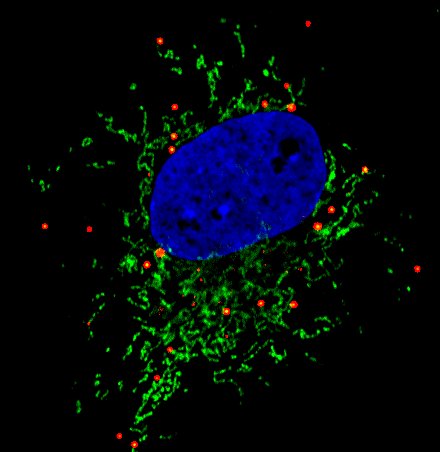
Most cells store neutral lipids in an organelle called a lipid droplet. Lipids are released from lipid droplets and used for energy metabolism or to build membranes. However, cells in the brain increase lipid droplets under pathological conditions. Our lab seeks to uncover the mechanisms and consequences of lipid droplet formation in neurodegenerative disease and stroke.

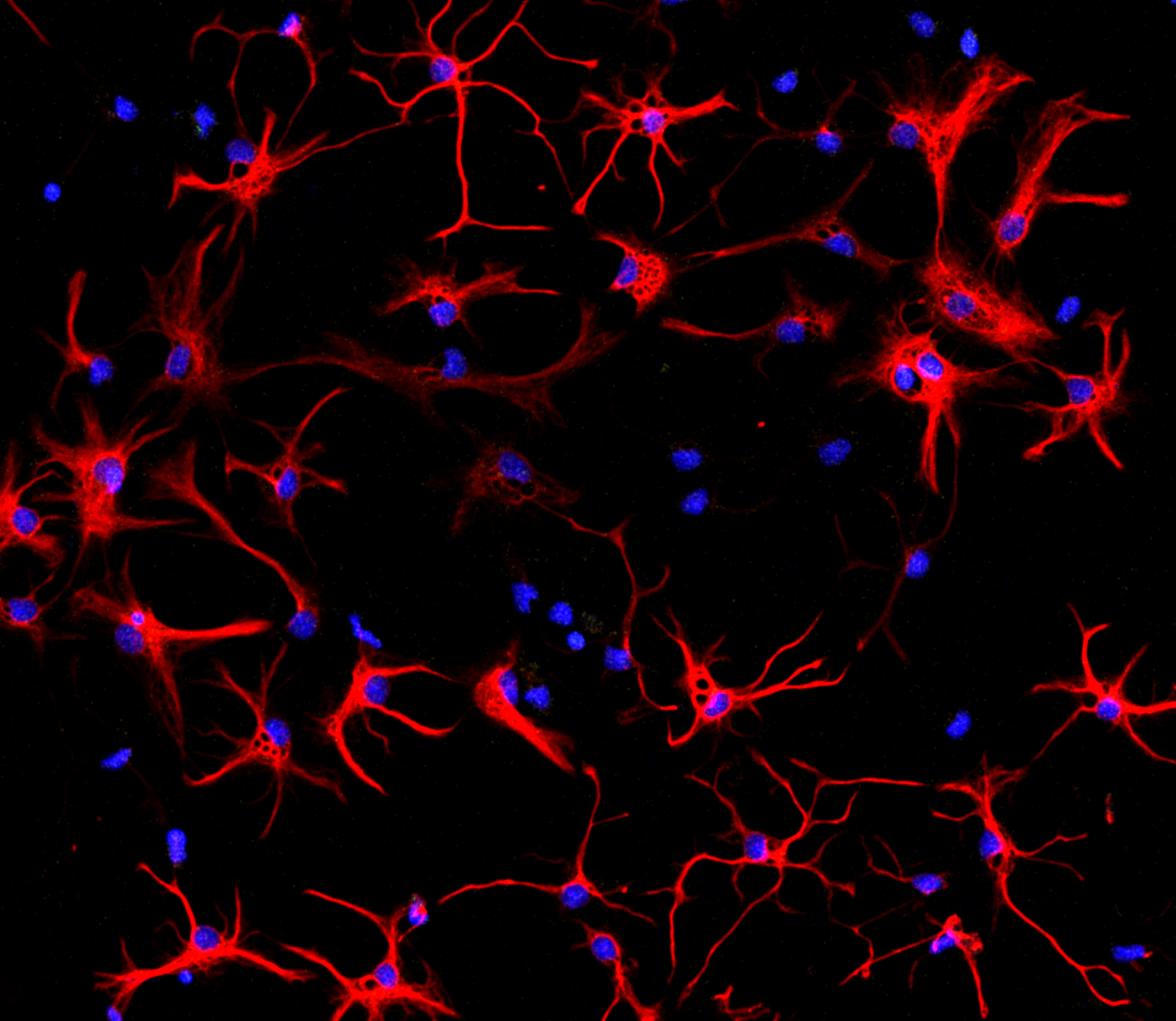
3. Lipid metabolism in neuron-astrocyte communication
Neuron-astrocyte communication is critical for the brain to function. The brain is abundant in lipids yet neurons have a limited capacity for lipid metabolism. Astrocytes on the other hand receive excess lipids from neurons and can readily metabolize them. Our lab is interested in the relationship between lipid metabolism and neuron-astrocyte coupling; more specifically, how does lipid metabolism shape neuron/astrocyte activity.

We are extremely grateful to the following funding agencies for their support: